 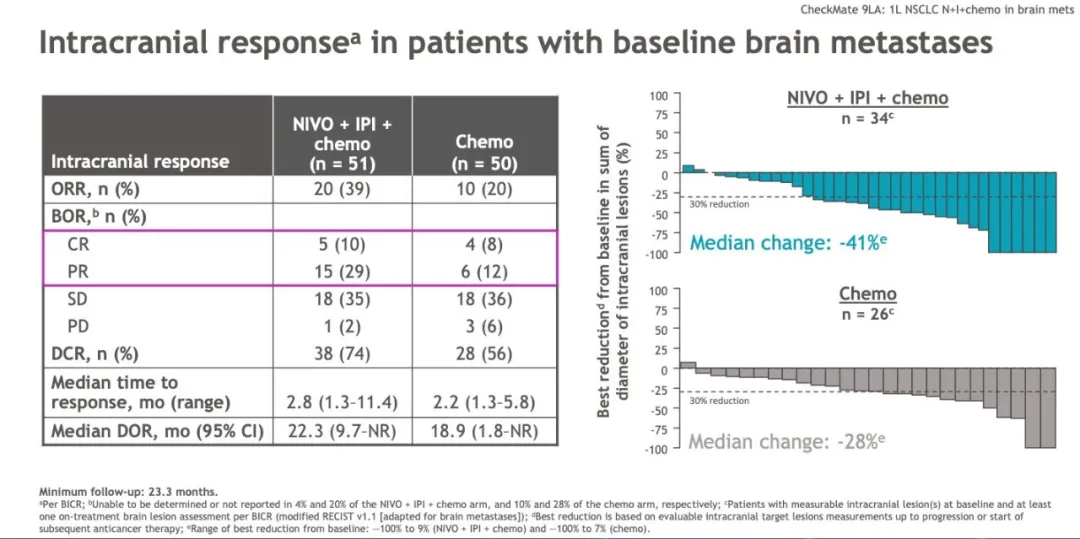
Addition of Ipi to Nivo also resulted in higher pCR rates, less viable tumor and enhanced tumor immune infiltration 8. We found that Nivo and Nivo+Ipi produced MPR rates of 22% and 38%, respectively. In the phase 2 randomized NEOSTAR study, we evaluated neoadjuvant Nivo or Nivo+Ipi followed by surgery in 44 patients with operable NSCLC 8. CheckMate-816 was the first large-scale phase 3 randomized trial to evaluate neoadjuvant nivolumab plus chemotherapy (Nivo+CT) versus chemotherapy (CT) alone in patients with resectable stage IB–IIIA NSCLC and demonstrated a pCR rate of 24.0% with Nivo+CT compared to 2.2% with CT alone, as well as improved event-free survival (EFS) 3, which led to FDA approval of neoadjuvant Nivo+CT as the new standard of care for patients with resectable NSCLC.Īnother strategy to enhance the efficacy of neoadjuvant anti-PD-(L)1 therapy is to combine it with the cytotoxic T-lymphocyte-associated protein (CTLA-4) immune checkpoint inhibitor ipilimumab (Ipi), given that the two inhibitors impact the immune system through two independent, and possibly complementary, mechanisms of action 12, 13. The addition of platinum-based chemotherapy to immunotherapy has proved promising 9, with initial phase 2 studies producing MPR and pCR rates of 57–83% and 33–63%, respectively 10, 11. Studies of neoadjuvant single-agent anti-PD-(L)1 therapy have yielded MPR rates between 6.7% and 45% 4, 5, 6, 7, 8. These trials have benefited by using major pathologic response (MPR) or complete pathologic response (pCR) as surrogate endpoints of long-term outcomes. Neoadjuvant immunotherapy trials are based on the premise that an intact tumor immune microenvironment elicits the most robust responses to immune checkpoint inhibitors 1. Now, considerable effort is focused on how to best leverage immune checkpoint therapy for patients with resectable early-stage NSCLC and prevent post-operative tumor recurrence 1, using adjuvant 2 or neoadjuvant 3 approaches targeting the PD-(L)1 axis. Immune checkpoint therapy has changed the treatment paradigm for patients with non-small cell lung cancer (NSCLC) however, until recently, much of the progress had been confined to the locally advanced and metastatic setting. 
Neoadjuvant Ipi+Nivo+CT enhances pathologic responses and warrants further study in operable NSCLC. Baseline fecal microbiota in patients with MPR were enriched with beneficial taxa, such as Akkermansia, and displayed reduced abundance of pro-inflammatory and pathogenic microbes. Single-cell sequencing and multi-platform immune profiling (exploratory endpoints) underscored immune cell populations and phenotypes, including effector memory CD8 + T, B and myeloid cells and markers of tertiary lymphoid structures, that were preferentially increased in the Ipi+Nivo+CT cohort. No new safety signals were observed in either arm. In patients without known tumor EGFR/ ALK alterations, MPR rates were 41.2% (7/17) and 62.5% (10/16) in the Nivo+CT and Ipi+Nivo+CT groups, respectively. MPR rates were 32.1% (7/22, 80% confidence interval (CI) 18.7–43.1%) in the Nivo+CT arm and 50% (11/22, 80% CI 34.6–61.1%) in the Ipi+Nivo+CT arm the primary endpoint was met in both arms. Here we report the results and correlates of two arms of the phase 2 platform NEOSTAR trial testing neoadjuvant Nivo+CT and Ipi+Nivo+CT with major pathologic response (MPR) as the primary endpoint. The impact of adding ipilimumab to neoadjuvant Nivo+CT is unknown. Neoadjuvant ipilimumab + nivolumab (Ipi+Nivo) and nivolumab + chemotherapy (Nivo+CT) induce greater pathologic response rates than CT alone in patients with operable non-small cell lung cancer (NSCLC). Nature Medicine volume 29, pages 593–604 ( 2023) Cite this article Neoadjuvant chemotherapy plus nivolumab with or without ipilimumab in operable non-small cell lung cancer: the phase 2 platform NEOSTAR trial 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
